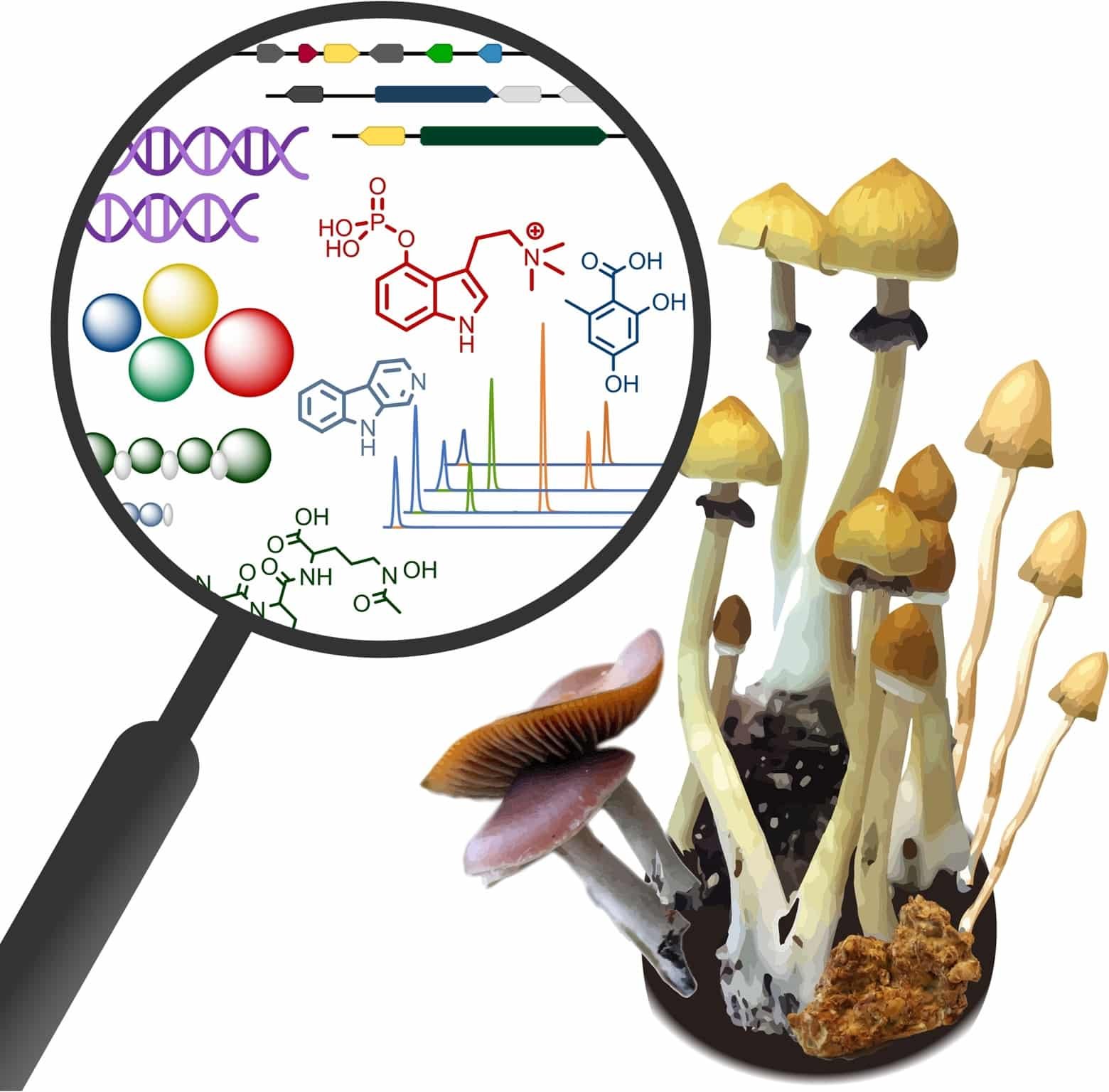
Ethanol is commonly used as a solvent in the extraction of bioactive compounds from plant and fungal materials, including psilocybin-containing mushrooms. Due to its polarity, relative safety, and food-grade availability, ethanol is particularly effective at dissolving compounds like psilocybin and psilocin for analytical, research, or educational purposes.
Note: Psilocybin is a controlled substance in many countries. Extraction or possession may be illegal depending on local laws. This content is provided for informational and scientific context only.
Sections
ToggleWhy Ethanol Is Used

Ethanol (ethyl alcohol) is favored in extraction processes for several reasons:
1. Polarity
Psilocybin is a polar compound, meaning it dissolves well in polar solvents. Ethanol, especially when mixed with water (e.g., 70–95%), effectively extracts polar alkaloids.
2. Relative Safety
Compared to other organic solvents (such as methanol or chloroform), ethanol is less toxic and safer to handle, particularly in food-grade form.
3. Accessibility
Ethanol is widely available and commonly used in herbal tincture preparation, making it practical for laboratory and botanical extraction contexts.
How Ethanol Functions in the Extraction Process
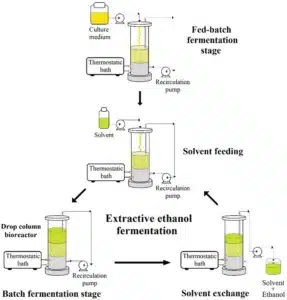
“In research contexts, ethanol serves several chemical roles based on its well-documented solvent properties found in analytical chemistry and its ability to dissolve polar molecules such as psilocybin.”
- Dissolution of Target Compounds
Ethanol interacts with the molecular structure of psilocybin, allowing it to transition from solid biological material into liquid solution. - Broad-Spectrum Solvent Action
Ethanol does not exclusively dissolve psilocybin. It may also extract:- Other alkaloids
- Organic acids
- Pigments
- Sugars
- Secondary metabolites
- Other alkaloids
- Preparation for Analytical Testing
Ethanol-based extracts are often used as samples for:- High-performance liquid chromatography (HPLC)
- Mass spectrometry
- High-performance liquid chromatography (HPLC)
Quantitative compound analysis
Stability and Handling Considerations
Psilocybin and related compounds can be sensitive to environmental factors. In research settings, attention is given to:
- Heat exposure
- Light (especially UV radiation)
- Oxygen
- Extreme pH conditions
Proper storage and controlled environments help maintain compound stability during analytical procedures.
Limitations of Ethanol Extraction
- Non-Selective Extraction
Ethanol may dissolve multiple compounds, requiring additional purification steps in laboratory analysis. - Variability in Concentration
Without analytical testing, solvent extracts cannot reliably indicate precise compound concentration. - Regulatory Restrictions
Because psilocybin is controlled in many regions, research use typically requires licensing or regulatory approval.
Conclusion
Ethanol is used in psilocybin extraction primarily because of its polarity, safety profile, and compatibility with laboratory analysis. It functions as a solvent that dissolves psilocybin from fungal material, often as part of broader analytical or pharmacological research. While effective, ethanol extraction is non-selective and must be paired with appropriate scientific methods for purification and quantification.